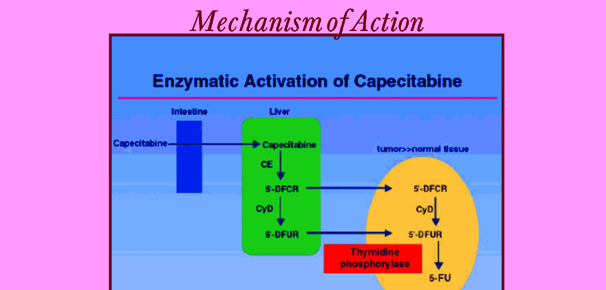
Cabdian
Indications :
Colon Cancer
-
Indicated as a single agent for adjuvant treatment in patients with Dukes’ C cancer.
- Indicated as a single agent for adjuvant treatment in patients with Dukes’ C colon cancer.
Breast Cancer
-
In combination with docetaxel for the treatment of patients
with metastatic breast cancer after failure of prior
anthracycline-containing chemotherapy.
- Monotherapy is also indicated for the treatment of patients with metastatic breast cancer resistant to both paclitaxel and an anthracycline-containing chemotherapy regimen.
Dosages: 1250mg/m2 P0 BD for 14 days
1000mg/m2 P0 BD for 14days in colorectal
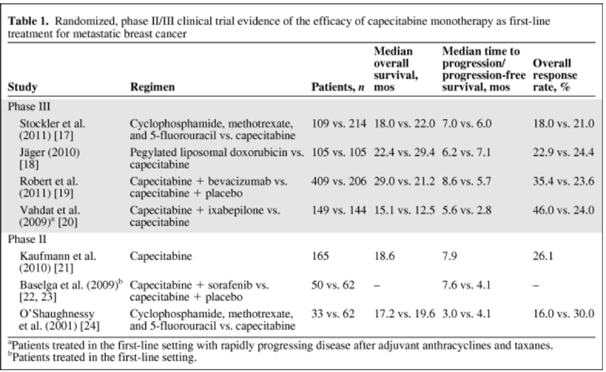
Expert's View
- Capecitabine is an effective, safe, convenient, and economically viable alternative to 5-FU as a first-line treatment for Metastatic Colorectalv Cancer.
-
Single-agent capecitabine maintenance therapy may be a potential treatment
strategy for MBc patients who responded to capecitabine-based chemotherapy
- Capecitabine could replace fluorouracil in adjuvant or neoadjuvant chemoradiotherapy regimens for patients with locally advanced rectal cancer.
-
CAPTEM is highly active, well tolerated and may prolong survival in patients with well-differentiated, metastatic NET who have progressed on previous therapies.
- Adjuvant treatment with capecitabine plus oxaliplatin after D2 gastrectomy should be considered for patients with operable stage II or III gastric cancer.
- Capecitabine, with or without oxaliplatin, could be considered as an alternative treatment option for elderly patients with stage III CRC .
- Capecitabine-based chemotherapy strategies show prolonged OS and enhanced ORR compared with traditional 5-FU-based treatments and therefore should be considered as one of the first choices for treatment for AGC. Asian patients also showed less grade 3 or grade 4 gastrointestinal toxicity with the capecitabine-based regimens.
- Single-agent capecitabine maintenance therapy may be a potential treatment strategy for MBC patients who responded to capecitabine-based chemotherapy.
- After initial disease control was achieved with the XT (capecitabine plus docetaxel) combination chemotherapy, capecitabine MT can significantly prolong PFS time with a favorable safety profile in mTNBC (metastatic triple-negative breast cancer) patients.